Leads To Development
Created in 2009

- BETA

Up & running (A)
Existing signals show a regular activity Social networks
981Activities
Technologies
Entity types
Location
202 Rue de la Croix Nivert, 75015 Paris, France
Paris
France
Employees
Scale: 11-50
Estimated: 20
SIREN
513398164Engaged corporates
1
0 1Added in Motherbase
3 years, 2 months ago
Value proposition
Nonclinical drug development solutions
LEADS TO DEVELOPMENT (L2D) provides expert management support for the early stage development of novel therapeutic agents, from initial concept and the design of a customised development plan through to writing and submission of the clinical trial applications.
Our services allow our clients to reach clinical milestones rapidly and cost effectively, without building unnecessary headcount and infrastructure. We assess project potential, design development strategies and deliver effective project advancement in order to minimise risk and increase project value.
Our experience of the biotechnology sector also allows us to offer a range of business services and strategic advice to investors and drug developers, including due diligence, drug positioning and project review.
Project management:
We provide operational management services for early stage therapeutic development. We can manage entire development projects or our services can be accessed on a punctual basis.
Consultancy, strategic & business services:
Our cumulated experience in the biotechnology industry also allows L2D to offer investors and key managers a range of strategic and business services and advice.
Our services provide the additional resources, proven experience and reliability required to reach the challenging milestones that often accompany early stage development programmes.
Consultancy, Operational project management, Preclinical development, and BiotechnologyOriginal language
Nonclinical drug development solutions
LEADS TO DEVELOPMENT (L2D) provides expert management support for the early stage development of novel therapeutic agents, from initial concept and the design of a customised development plan through to writing and submission of the clinical trial applications.
Our services allow our clients to reach clinical milestones rapidly and cost effectively, without building unnecessary headcount and infrastructure. We assess project potential, design development strategies and deliver effective project advancement in order to minimise risk and increase project value.
Our experience of the biotechnology sector also allows us to offer a range of business services and strategic advice to investors and drug developers, including due diligence, drug positioning and project review.
Project management:
We provide operational management services for early stage therapeutic development. We can manage entire development projects or our services can be accessed on a punctual basis.
Consultancy, strategic & business services:
Our cumulated experience in the biotechnology industry also allows L2D to offer investors and key managers a range of strategic and business services and advice.
Our services provide the additional resources, proven experience and reliability required to reach the challenging milestones that often accompany early stage development programmes.
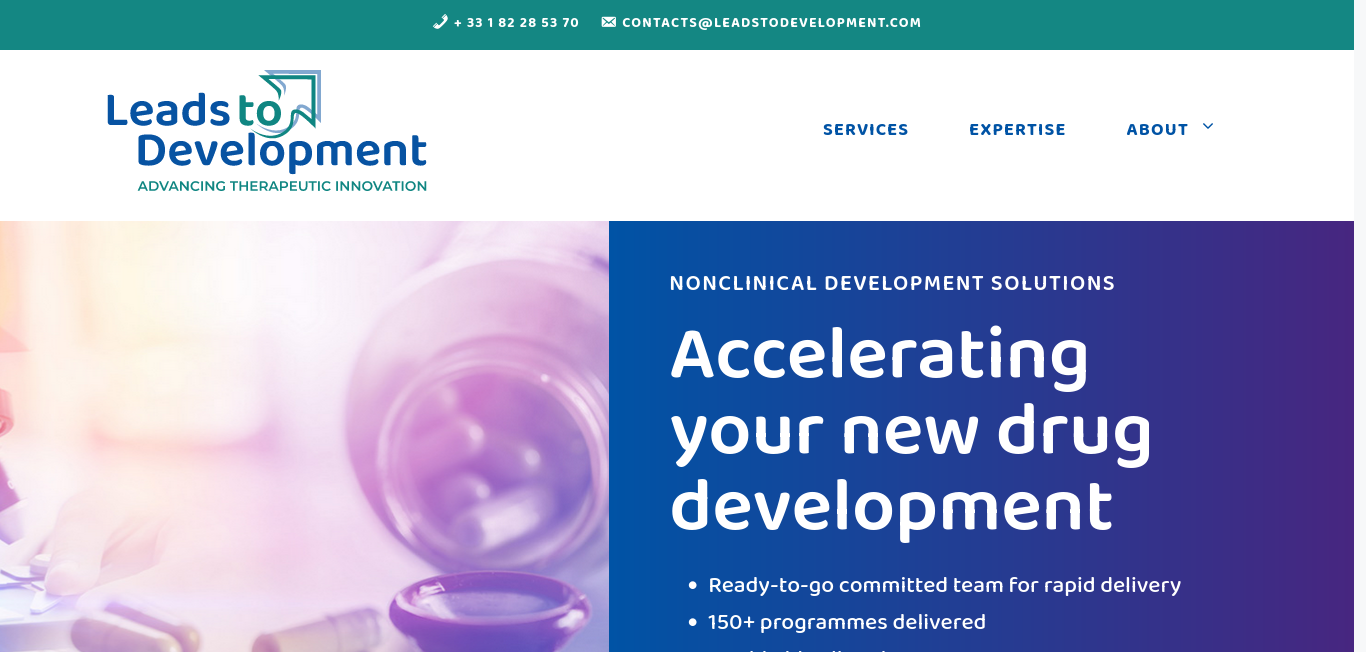
Nonclinical, CMC & Regulatory Drug Development Consulting Services
Nonclinical, CMC & Regulatory Drug Development Consulting Services. Accelerating your new drug development. Ready-to-go committed team for rapid delivery. 150+ programmes delivered.
https://leadstodevelopment.com/
| Corporate | Type | Tweets | Articles | |
|---|---|---|---|---|
 Institut Curie Health, Hospitals and Health Care | Institut Curie Health, Hospitals and Health Care | Other 24 Nov 2022 | | |